Richard Denison, Ph.D., is a Senior Scientist.
Last week, the National Academy of Sciences (NAS) held a joint meeting of its two panels that are charged with reviewing the listings of formaldehyde and styrene as carcinogens in the 12th Report on Carcinogens, which was released in June 2011.
The 12th Report on Carcinogens (RoC) is the latest edition of a Congressionally mandated report developed by the National Toxicology Program (NTP). It upgraded formaldehyde to the status of “known to be a human carcinogen,” and for the first time listed styrene as “reasonably anticipated to be a human carcinogen.” That put the chemical industry into a real tizzy, what with the threat these listings pose to its profits from the huge volumes of these cash cows sold each year, not to mention the huge potential liability it faces.
Never one to go down lightly, the American Chemistry Council (ACC) has launched an all-out assault on the NTP and the RoC. It is waging battle not only with the executive branch, but also in the courts and in Congress. In late 2011, it managed to get its allies in Congress to slip into the Consolidated Appropriations Act of 2012, without any debate, a rider that mandated the NAS reviews of the formaldehyde and styrene listings in the 12th RoC that are now underway.
ACC also pushed legislation in the last Congress to shut down all funding for the RoC until the reviews are completed; failing on that front, earlier this month it demanded that NTP cease all work on the next (13th) edition of the RoC. (For more background, see previous blog posts by EDF and NRDC.)
Lost in all this kerfluffle, however, are these salient facts:
- The formaldehyde and styrene listings are the outcome of one of the most extensive scientific assessment processes on the planet, entailing reviews by four separate groups of expert scientists for each chemical.
- ACC as well as the public had at least three separate formal opportunities for providing input to these expert bodies.
- Of a total of 52 votes cast by these scientific panels on the NTP’s recommended listings, 51 of those votes supported the recommendations and only one opposed them.
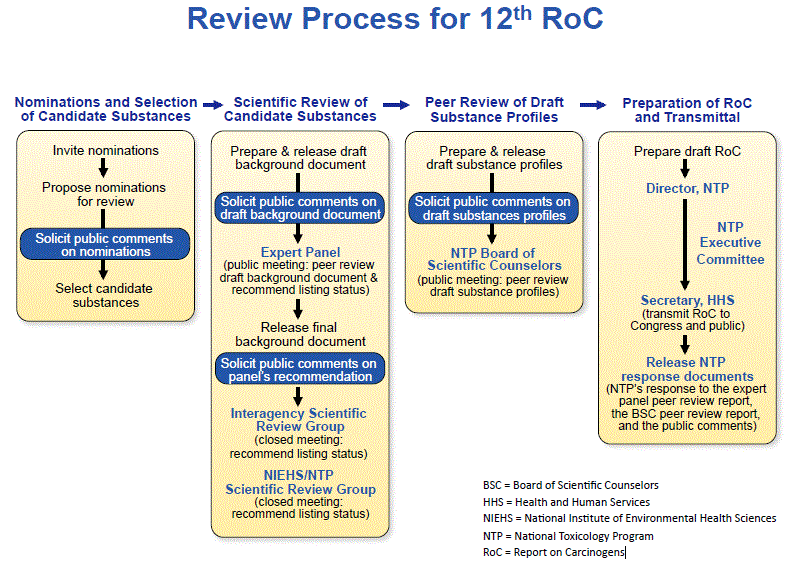
At last week’s joint panel meeting, Dr. John Bucher, head of the NTP, spent two hours laying out the process NTP uses to: (a) determine which chemicals get selected for possible inclusion in the RoC, (b) compile and assess the evidence to reach recommendations on the potential for those chemicals to cause cancer in humans, (c) solicit and consider public comments at each stage of the process, and (d) peer review NTP’s recommendations, first, by independent scientific experts, then by scientists drawn from multiple agencies across the federal government, and finally by another group of independent experts, the NTP’s Board of Scientific Counselors.
Here’s a schematic that summarizes NTP’s process; note the multiple rounds of scientific review and solicitation of public comment:
And here are the vote tallies from the expert groups:
- Recommendation to list formaldehyde as “known to be a human carcinogen”
- Expert panel: 9 yes to 0 no
- Interagency scientific review group: 8 yes to 0 no
- NIEHS/NTP scientific review group: 9 yes to 0 no
- Recommendation to list styrene as “reasonably anticipated to be a human carcinogen”
- Expert panel: 10 yes to 0 no
- Interagency scientific review group: 8 yes to 0 no
- NIEHS/NTP scientific review group: 7 yes to 1 no
For those of you keeping score, that’s 51 yes votes to 1 no vote.
Interestingly, two members of the interagency scientific review group for styrene actually thought that NTP’s recommendation was too weak and should be upgraded to “known to be a human carcinogen.”
None of this virtual unanimity among scientific experts has had any discernible effect on the hysterics of the chemical industry: It dominated the NAS’s joint panel meeting, flying in its own “experts” from all over the country. Of 11 public comments provided to the panelists, eight were from paid industry consultants or staff from ACC or its members companies.
Bracketing these loud industry voices, however, were a couple others. The first public comment of the day came from the president and founder of a small personal care products company that is a member of the American Sustainable Business Council (ASBC). She noted how critical it is for small businesses making consumer products to have access to timely, objective information on the hazards of chemicals, so that they can take steps to reduce or eliminate them. She also pointed to a recent national poll of small business owners that found widespread support – regardless of party affiliation – for greater access to information about toxic chemicals and stricter regulation.
My colleague at NRDC, Dr. Jennifer Sass, and I were the last two public commenters of the day. In her comments, Dr. Sass asked the obvious question: “Why is the [NAS] reviewing the styrene and formaldehyde assessments, which have both already undergone years of public and scientific review?” She also provided critical rebuttal to the industry’s claims that NTP somehow got it wrong.
I made two points in my comments: First, delays in reaching decisions about toxic chemicals – in this case spurred by the chemical industry’s political attack on independent government science – have real-world consequences: They mean we continue to needlessly expose millions of people to these chemicals’ harmful effects. Industry interference has kept the federal government from completing its assessment of formaldehyde’s risks for 15 years and counting. Had those delays not occurred, we might well have avoided the national scandal that arose when victims of Hurricane Katrina were poisoned after being housed in those infamous FEMA trailers that were laced with formaldehyde-emitting plywood.
Second, I urged the members of the NAS panels, when carrying out their charge to ensure the science behind NTP’s listings is sound, to avoid demanding “perfect” science and raising the bar so high before government can act that they inadvertently contribute to even more delay in securing critical health protections for workers, people of color and lower socioeconomic status and others who disproportionately bear the impact of toxic chemical exposures.