Richard Denison, Ph.D., is a Senior Scientist.
[Earlier posts in this series can be found here and here.]
Over the past decade, the Environmental Protection Agency (EPA) has pursued a voluntary program, the High Production Volume (HPV) Chemical Challenge, as a means to fill the enormous gaps in publicly available data on the hazards of the most widely used chemicals in the U.S. Using the Challenge data, EPA has recently begun assessing HPV chemicals under its Chemical Assessment and Management Program (ChAMP). But is ChAMP up to the job?
While the HPV Challenge has fallen well short of its promise to ensure timely development and public access to high-quality hazard data on HPV chemicals, it has provided EPA with a greater ability to screen such chemicals for potential hazards than was previously possible. That’s where ChAMP enters.
ChAMP grew out of an overly-ambitious U.S. commitment made in August 2007 under the North American Security and Prosperity Partnership (SPP). ChAMP commits EPA to assess about 2,750 HPV chemicals as well as about 4,000 medium production volume (MPV) chemicals by 2012.
Last year, EDF provided to EPA a critique of ChAMP, raising numerous serious concerns about the direction it was taking. Among them was a lack of transparency about the extent of data and methodology it was using in its assessments.
The good news is that EPA recently issued several documents that significantly enhance the transparency of its assessment process.
The bad news, unfortunately, is that these documents make clear that EPA is intent on pursuing – now at breakneck speed – a scientifically flawed approach to assessing HPV chemicals under ChAMP.
In this and subsequent posts, we will update and expand our critique and provide a series of specific examples that illustrate the unsupportable risk decisions EPA is making under ChAMP.
What does EPA do under ChAMP?
To provide a good understanding of ChAMP, I need to walk through some background and introduce some arcane terminology:
Hazard characterizations
EPA began issuing so-called “hazard characterizations,” or HCs, starting in September 2007, and has now posted HCs covering several hundred HPV chemicals. This step was called for by a federal advisory committee (on which EDF sat) that EPA convened to assist it in deciding what to do with the HPV Challenge hazard data it was compiling. As the committee requested, EPA agreed to do the following for each HPV chemical:
- conduct an objective evaluation of the quality and completeness of the data set in the HPV Challenge Program submission;
- independently assess the submitted data for each endpoint to determine the level of hazard of the substance; and
- develop and make public a hazard characterization of the substance(s).
As EPA’s HCs have been made public, serious concerns have emerged over both the quality and completeness of data for many HPV chemicals. EPA’s own accounting indicates that data gaps remain in about 30% of the supposedly final HPV submissions it has reviewed, and our analysis has identified even more.
The very foundation of the HPV Challenge was that companies were to provide a hazard data set specified through an international consensus process as the minimum amount of data needed to conduct even a screening-level hazard assessment for a chemical. The fact that serious data gaps remain calls into question, therefore, EPA’s ability to characterize hazards of these chemicals even at a screening level.
So how has EPA responded to these data gaps?
- To date, EPA has not initiated any regulatory action to require that these gaps be filled, despite a promise to do so when it launched the HPV Challenge.
- EPA has frequently obscured or glossed over the extent of missing data, although more recent postings have been more transparent in this regard.
- In the few high-priority cases it has identified (more on this below), EPA’s only response to data gaps has been to “encourage companies to provide available information on a voluntary and non-confidential basis.”
- Moreover, even for chemicals with data gaps, EPA is routinely characterizing almost all of them as of low or moderate hazard – an unsupportable conclusion on policy and scientific grounds.
It would be one thing for EPA to identify as high-hazard those chemicals where, despite the data gaps, available data demonstrate high toxicity. It’s quite another for EPA to effectively exonerate chemicals as low-hazard when not even a bare-minimum data set is available for them.
Risk-Based Prioritizations
Although not required by the SPP agreement or called for by its advisory committee, EPA has on its own expanded its commitment to assess the risk, not just the hazard, of all HPV chemicals by 2012.
As will be explained below and in subsequent posts, this rash decision further strains the credibility of ChAMP. EPA is quite literally rushing to crank out what it calls “risk-based prioritizations,” or RBPs, for hundreds of HPV chemicals. These documents rank the chemicals as being of low-, medium- or high-priority from a risk perspective. But once again, EPA is basing its rankings on data that fall far short of what would be needed to do a credible screening-level assessment of risk.
To assess risk, one needs to have information about the use of and exposure to a chemical, as well about its hazards. Remember the old equation:
hazard x exposure = risk
EPA had high hopes – I would say unrealistically high – that it would get good use and exposure information through something called the Inventory Update Rule, or IUR. Starting in 2006, manufacturers of HPV chemicals were required to report certain information about the downstream processing and uses of their chemicals, which is relevant to ascertaining the potential for exposure of the public, workers, consumers and children, as well as the environment (see Section VI of this paper for details).
EPA’s idea was that it could combine the HPV Challenge hazard data with the 2006 IUR processing and use data, and draw conclusions about how risky HPV chemicals are.
Reliance on poor chemical use information
I’ve already summarized some of the many limitations of EPA’s hazard data. The situation with the quality and completeness of the IUR processing and use information is far worse.
Consider the limitations to the IUR reporting program itself:
- IUR reporting is limited to only a single year in each 5-year reporting cycle, so currently EPA is relying only on data reported for 2005.
- Only manufacturers are required to report under the IUR, not downstream processors, distributors or users of the chemicals.
- Manufacturers can claim any and all of the information they submit to be confidential business information (CBI); while EPA can see the CBI, the public cannot. According to EPA, about a third of the processing and use data were claimed to be CBI; see Exhibit 3 in this report.
- When EPA developed the reporting requirements, it required manufacturers to report processing and use information only to the extent it was readily obtainable. So how often did the companies maintain such data were not readily obtainable? Often enough that EPA actually coined a new acronym – NRO – for such situations. EPA’s summary IUR reportindicates that manufacturers made NRO claims:
- about 10-15% of the time, for the processing data elements (e.g., type of process applied to the chemical or in which it is used, industrial function of the chemical),
- about 20% of the time, for information about the commercial or consumer product categories in which their chemicals were used, and
- a whopping 40% of the time, when indicating whether the chemical was known to be used in products intended for use by children.
It is widely acknowledged that manufacturers are often in a very poor position to know much about the downstream processing and use of the chemicals they produce. See, for example, here and here. Even chemical manufacturers agree: the Society of Chemical Manufacturers and Affiliates (SOCMA), a chemical industry trade association, cited this concern when it filed comments vigorously opposing EPA’s addition of processing and use information to the IUR in the first place. No doubt that helped them to get what has turned about to be a huge loophole in the reporting requirements.
So it’s not surprising that EPA received only limited data on HPV chemicals through its IUR reporting. The real question is why does EPA think such incomplete data derived solely from chemical manufacturers is sufficient to draw definitive exposure and risk conclusions about HPV chemicals?
Here again, it would be defensible for EPA to use incomplete use data to identify chemicals posing a high exposure potential, if the available data so demonstrate. It’s quite another for EPA to set aside chemicals as low-exposure (and hence low-risk) based on such sketchy data.
What does it take to get flagged under ChAMP as a chemical of concern?
So we have established that EPA is using often-incomplete data to rank HPV chemicals as:
- high/moderate/low hazard and
- high/medium/low exposure,
and then it combines these to rank the chemicals as high/medium/low risk.
One might reasonably assume that – given the decidedly mixed quality and completeness of the data EPA is using – it would err on the side of caution so as not to prematurely eliminate potentially risky chemicals. After all, this whole exercise is a “screening” process, designed to flag chemicals for further scrutiny: Including “false positives” – chemicals initially thought to be of concern that turn out not to be – is of far less concern than excluding “false negatives” – chemicals that actually are of concern. That’s because any false positives will be detected later upon further examination, whereas false negatives set aside won’t be looked at further and hence won’t be discovered to have been incorrectly classified.
Here’s what EPA is actually doing, however:
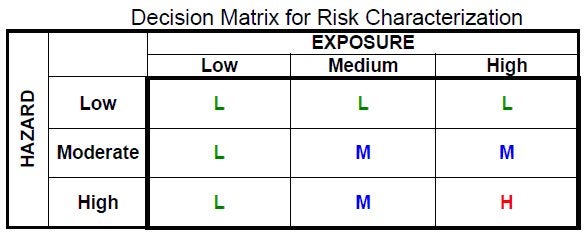
A prudent approach to screening would be to acknowledge that a high-hazard chemical ought to have awfully strong evidence of low exposure before relegating it to the low-risk dustbin. Likewise, one ought to have a pretty high degree of confidence that a chemical to which exposure is expected to be high is low-hazard before deciding it can be set aside.
The decision matrix for a prudent screening system would look like this:
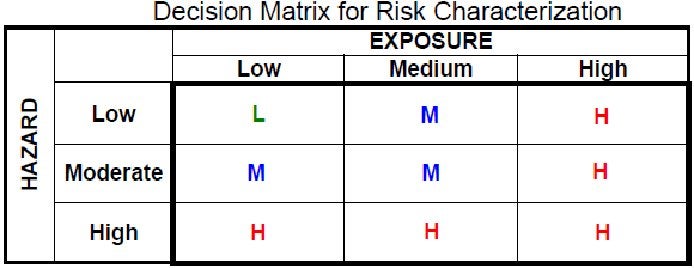
or at least like this:
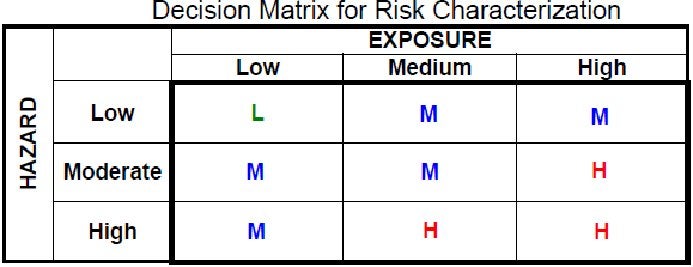
But no: EPA acts as if the information it’s using is complete and perfect. How else could it possibly support a screening approach that says: no matter how hazardous a chemical is, we’ll only consider it high-risk if we are quite certain exposure to it will occur.
At this point, it probably won’t surprise you to learn that, of 212 chemicals for which EPA has issued RBPs, 6 chemicals were ranked high-risk, 56 medium-risk, and 150 low-risk. (To its credit, EPA has also ranked eight more chemicals as high-priority, because their sponsors never followed through on their HPV Challenge commitments, and hence the chemicals lack hazard data altogether.)
EDF is not alone in flagging the fact that EPA’s approach to risk ranking is the opposite of health-protective. EPA’s own Children’s Health Protection Advisory Committee (CHPAC) raised very similar concerns about ChAMP in a letter sent to the EPA Administrator last year.
EPA will at least aggressively seek better information, right?
Well, no. The irony and real tragedy of EPA’s decision logic under ChAMP are that the hundreds of chemicals it relegates to low-priority status will likely never be targeted for development of better information on which to more accurately assess their risks. That’s because EPA clearly states there is to be “no follow-up action at this time.” See, for example, here and here.
Even for medium- and high-priority chemicals, the most EPA can muster in the way of any action to address data limitations is to say that “companies are encouraged to provide available information on a voluntary and non-confidential basis.” See, for example, here and here.
Conclusion
I’ll sum up this post as follows: Under ChAMP, EPA has constructed a house of cards, piling marginal data on top of questionable assumptions on top of a decision logic that is the antithesis of a prudent screening approach. The result is a system that is so rigged against identifying a chemical to be of high potential risk that – voila! – virtually none are being found!
It’s getting harder and harder to believe that this is all an accident. The chemical industry has for years pleaded with EPA not to publicize chemical hazards without “putting the information into a risk context,” lest it scare the public. ChAMP seems to be fulfilling industry’s every wish, supplying the risk context in a manner that bends over backward to argue that even high-hazard high-volume chemicals are of low priority for further scrutiny – and in the process preclude even getting better information about such chemicals.
ChAMP is a holdover from the last Administration. Will it become EPA’s “gift that keeps on giving” to the chemical industry?
Many of the shortcomings of the ChAMP initiative – most notably its reliance on incomplete and often questionable data to “prioritize” chemicals (see my earlier post on the debate over what prioritization means) – can be directly traced to structural limitations in the authority EPA has under the Toxic Substances Control Act (TSCA), which restrict EPA’s ability both to identify and act to control dangerous chemicals. This is especially ironic, since ChAMP is being actively positioned as either obviating the need – or providing a model – for TSCA reform. See examples here, here and here.
So going forward, we’ll also put the direction EPA has chosen for ChAMP and the wholesale chemical industry endorsement of it into the context of the broader debate around TSCA reform.
Coming next: This is the first of several posts that aim to expose the enormous problems with the direction EPA has taken ChAMP. Over the coming weeks, we’ll provide a more specific critique and analysis of ChAMP’s RBPs, and we’ll start posting individual case examples of chemicals that directly illustrate the manner in which EPA has managed to prematurely exonerate hundreds of HPV chemicals.
Come back soon.










One Comment
please add me to your email push list